Introduction
A 32-year-old man was admitted for passing out twice in a shopping mall. Following investigation, he proved to have suffered substantial bilateral pulmonary embolism (PE) from an aneurysm of his right popliteal vein. While on therapeutic systemic heparin therapy, his physician asked for a filtering device in his inferior vena cava, but was advised against this by a consulting specialist because of the patient’s youth and potential complications and long-term morbidities that caval filtration may incur. The patient eventually received an optional IVC filter (vide infra) and turned out on later workup to also have subacute thrombosis of his right popliteal artery and its branches. These and the thrombosis of the popliteal vein were cleared with catheter-directed thrombolysis and the patient went home well while systemically anticoagulated. He awaits surgical repair of the aneurysm of the popliteal vein while undergoing workup for thrombophilia. This event illustrates how disparate decisions on caval filtration can be, in part a fall-out of the paucity of randomized controlled trials on the therapy and it spawned in my mind the questions I address in this writing.
Images
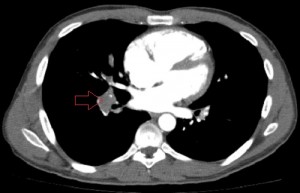
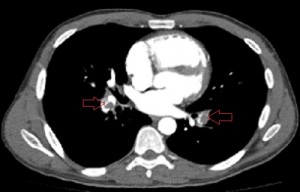
Axial images of CT angiogram of the pulmonary arteries 1 day after the patient’s admission showing filling defects in the central branches of both pulmonary arteries.
What is the role of caval filters in the management of venous thromboembolism (VTE)?
The sole purpose of a filtering device in the inferior vena cava is to prevent pulmonary embolism. Caval filtering devices do not reverse or prevent venous thromboembolism, whose primary therapies are pharmacologic or mechanical. Systemic anticoagulation prevents further formation of clot, allowing native (intrinsic) fibrinolysis to clear or recanalize formed thrombus. When the burden of deep vein thrombosis (DVT) is substantial or the patient symptomatic from it, synthetic thrombolytic agents, with or without mechanical disruption of the clot, are employed to clear the clot or at least debulk it. Anticoagulation and external mechanical compression are also employed to prophylax against VTE, the former at much lower doses than when used therapeutically, that is, when clot is already formed. Symptomatic pulmonary thromboembolism (PE), the most serious complication of venous thromboembolism, occurs in 355,000 people in the USA every year and claims 240,000 lives for the same time period. Chronic or recurrent PE exerts great morbidity on its victims through chronic hypoxemia, pulmonary arterial hypertension and cor pulmonale; untreated, it ultimately kills. Emboli arrive the pulmonary circulation from the deep veins of the lower and upper extremities and the pelvic veins, journeying past the inferior and superior vena cavae on their way. Thus, although the primary treatment for and prevention of PE is anticoagulation, it is prudent to place a filtering device in the IVC in some class of patients with deep vein thrombosis.
What are the types of caval filters and what determines the type a patient receives?
Before discussing the indications for caval filtration, let us review the types of IVC filters currently available. Often, when I receive a request for caval filtration, it reads “Greenfield filter”. I suppose that this is because the Greenfield filter was the first in the generation of IVC filters placed percutaneously and, even though no longer available in its original design, became the benchmark against which newer filters are compared. Following its percutaneous placement in 1973, a quick succession of filters with trimmer delivery systems appeared on the market and today there are some 15 designs of IVC filters approved by the department of Food and Drug Administration (FDA) in USA. The designs of modern IVC filters allow classification into temporary, permanent and optional filters. Permanent filters are irremovable; once deployed they stay in the patient. They find use in patients whose indication(s) for caval filtration is (are) unlikely to go away, for example, terminally-ill patients unlikely to live beyond 6 months or elderly patients prone to falls and are unlikely candidates for systemic anticoagulation. There are 2 types of optional filters – removable (retrievable) filters and convertible filters. Filters removed or retrieved from patients when the risk for PE for which they were deployed passes are called removable or retrievable filters, while those whose construction allows their conversion into IVC stents are called convertible filters. Optional filters are ideal for patients whose need for caval filtration is brief, for example, young patients needing filters for short times, trauma patients who cannot be anticoagulated or patients undergoing surgical interventions that briefly heighten their risk of VTE and cannot be anticoagulated. There is another class of filters called temporary filters previously available in Europe but no longer in use today. They possessed intravenous components harnessed to external anchoring parts and were used for brief needs of caval filtration. Today, most IVC filters in the USA are either permanent or removable filters; there are no convertible filters. Optional and temporary filters reduce the complications associated with permanent caval filtration such as caval thrombosis and fracture and migration of the filter or its parts. Optional filters may be left in patients if the need arises, with no more adverse effects than are associated with permanent filters. Such may happen in a patient whose need for caval filtration, considered brief at the outset translates into an irreversible or permanent need. There are at present no special set of indications for optional IVC filters distinct from those for permanent filters except as dictated by the anticipated length of the need for caval filtration. Portions of a filter in contact with the caval wall are engrafted into the wall and if they include the hook or head of the filter, which is what is grabbed for retrieval, it makes the process difficult or impossible and the longer a filter dwells in the IVC, the more ensconced therein it will be. So, removable filters should be removed within the time frame recommended by their manufacturers or as close to the expiration date as possible. Generally, there is 100% rate of successful removal when they are removed within their suggested times for removal, which vary greatly and are components of “improvements” each new arrival to the market touts as a leverage over older designs. Removing retrievable filters past their recommended retrieval dates is feasible but more difficult, but some practitioners claim enviable successful removal rates regardless of the duration of implantation. In addition to engraftment into the venous wall, another reason for not retrieving removable filters is the loss of the patients to follow-up, either due to inadequate mechanisms for follow-ups or patients’ attitudes towards health care. All practitioners inserting removable filters should strive to have efficient mechanisms of getting their filters out of their patients.
What are the indications for IVC filter placement?
As I indicated above, the primary treatment for VTE is anticoagulation, not caval filtration whose principal role in the treatment of VTE is prevention of symptomatic PE. Therefore, DVT or a reasonable risk of its developing must exist in association with the presence of downside(s) to the institution of systemic anticoagulation before considering caval filtration. In other words, the risk of DVT and PE without a recognized downside to anticoagulation, or vice versa, is not sufficient reason for caval filtration; both must co-exist to justify caval filtration. The downsides to systemic anticoagulation are its contraindications (e.g., ongoing GI blood loss, etc.), complications (e.g., intracranial hemorrhage, frequent falls, etc.), ineffectiveness (e.g. breakthrough PE despite therapeutic anticoagulation, etc.), and unreliability or unpredictability (noncompliant or homeless patient, etc). These make the use of systemic anticoagulation to treat VTE unwise, risky and harmful and justify filtering the vena cavaThere are now three working categories of indications for caval filtration as shown in the table below.
|
Absolute indications (proven VTE) |
Relative indications (proven VTE) |
Prophylactic indications (no VTE, high risk for anticoagulation) |
|
Recurrent VTE despite adequate anticoagulation |
Iliocaval DVT |
Trauma patient with high risk of VTE |
|
Contraindication to anticoagulation |
Large, free-floating thrombus |
Surgical procedure in a patient a high risk of DVT |
|
Complication of anticoagulation |
Thrombolysis of iliocaval DVT |
Medical condition with high risk of DVT |
|
Inability to achieve or maintain anticoagulation |
VTE with limited cardiopulmonary reserve |
|
| Poor compliance with anticoagulation |
||
|
High risk of complication of anticoagulation
|
From Kaufman JA, Kinney TB, Streiff MB, at al. Guidelines for the use retrievable and convertible vena cava filters: report from the Society of Interventional Radiology multidisciplinary consensus conference. J Vasc Interv Radiology 2006;17.
Notice that patients with absolute and relative indications for caval filtration have proven VTE, whereas those with prophylactic indications do not. Caval filtration for absolute indications is firm-footed, while for relative and prophylactic indications, there is wiggle room for practitioners to vary in their decisions. Some will deploy filters and others will not, informed by experiences and choices. Thus, the practice of caval filtration varies with physicians and where they practice, but with the availability of optional filters there is rising tendency towards caval filtration for the duration of the expected risk and removing the filter when the risk passes. I find the following suggestions on the use of IVC filters useful. They come from the Society of Interventional Radiology’s consensus document on the guidelines for the use of retrievable and convertible filters published in 2006: 1.First, determine that a patient is at risk for clinically significant PE. If such risk does not exist, then there is no reason for caval filtration. 2.If the risk exists, determine if anticoagulation (AC) or primary prophylaxis is feasible. If it is, then do so; an IVC filter is not necessary. 3.If AC or prophylaxis is not feasible (contraindicated, unreliable, unpredictable, ineffective, or attended by complications), decide if the duration of the risk for PE and/or the downside to AC is short: 1. If it is, deploy an optional filter. 2. If it is long, deploy a permanent filter. 3. If it is uncertain, deploy either filter type.
What are the contraindications to caval filtration?
Some patients will decline caval filtration despite sound medical advice and should have no filter placed in them. There will be uncommon times when the natural paths to the inferior vena cava are thrombosed, fibrosed, ligated or otherwise render the IVC inaccessible. But it can be argued that filtering the vena cava at such times is a mute point because migrating clots have no straight path to the lungs. This is true except in instances when collateral paths of venous return become as large as or larger than the vena cava and may be conduits for successful pulmonary embolism. Unless unavoidable, permanent caval filtration should be avoided in adolescents because at the present there is not enough data on how this will turn out in the long run. Finally, filters should not be placed in thrombosed or fibrotic vena cava because the presence of clot or fibrotic tissue prevents proper apposition of the filter struts against the caval wall and limit full filter opening. These in turn reduce the efficiency of the filtering device and, in the case of a removable filter, may render the device irremovable.
Can filters be placed in the superior vena cava?
Yes, filtering devices can be and have been safely placed in the superior vena cava (SVC) to interrupt embolism of clot to the lungs from the upper limbs, although to a much lesser frequency than they are placed in the inferior vena cava. The determinants of SVC filter placement include the burden of upper extremity thrombosis, the patient’s pulmonary circulation’s existing reserve (can it safely tolerate new emboli?), and the availability of local skill for SVC filter placement. Often, however, VTEs of the upper extremities are primarily managed with systemic anticoagulation assisted by the application of external compressors and limb elevation. When patients are very symptomatic (painful, swollen limbs), the clot may be cleared or debulk pharmaco-mechanically.
If caval filters play such important role in preventing pulmonary thromboembolism, why are they not placed in all patients?
Although the placement of IVC filters is relatively safe, like all procedures it not free of risks and downsides. There may be complications during the filter placement such as bleeding at the access site, injury to the adjacent arteries, cardiac arrhythmias, deployment of the filter in a wrong vein such as the iliolumbar vein or a hepatic vein, perforation of the inferior vena cava, or migration of the filter to the heart which requires surgical extraction if not immediately fatal. In addition, particularly for filters left in place for long such as permanent filters, there is increased risk of deep vein and caval thrombosis compared to patients with VTE treated with systemic anticoagulation alone. This is the reason it is reasonable to anticoagulate patients with IVC filters when it is safe to do so since the 2 forms of therapy serve different purposes: one prevents formation of new clot, while the other prevents migration of formed clot to the lungs past the IVC but at the cost of increasing the risk of more clot formation. The findings of the French study “Prévention du Risque d’Embolie Pulmonaire par Interruption Cave, PREPIC”, the only RCT on the use of permanent IVC filters to prevent symptomatic PE, do not encourage flippant deployment of IVC filters in patients. The study compared the use of anticoagulation to treat PE to its use combined with a permanent IVC filter at 2 and 8 years. At 2 years there was more reduced risk of PE in patients treated with both anticoagulation and IVC filter placement (an occurrence rate of 3.4% versus 6.3% in those without an IVC filter). At 8 years these values rose to 6.2% versus 15.1%, respectively. But DVT developed in 20.8% of these patients compared to 11.6% of those without filters at 2 years, a difference that in 8 years rose to 35.7% versus 27.5%. It was thus clear, at least from this study, that permanent IVC filters are beneficial but carry a long-term risk of DVT that cannot be ignored. Their use must tempered by this knowledge. Though there are as yet no RCTs on the benefits and risks of optional filters, the relative brevity of their use is a favored reason for their explosive use in recent times in the prevention of PE, an explosion likely to continue.
Conclusion
The physician contemplating caval filtration but is baffled by the lack of sound guidelines on it application should recall that the primary method of treating and preventing VTE or DVT is anticoagulation, whereas placing a filtering device in the IVC reduces the risk of fatal or massive pulmonary artery embolism, a dreaded complication of VTE whose occurrence is difficult to forecast. But the use of IVC filters, though largely safe comes with a price that cannot be ignored, a price which the use of removable filters is attempting to overcome. For this effort to yield fruits, however, removable filters should be removed when they serve their need though they may be left in situ when the risk of removing them outweighs the risk of leaving them in the patient. The placement of an IVC filter does not eliminate the need for systemic anticoagulation in patients without absolute contraindication to their use because it reduces the risk of more DVT and caval thrombosis.
References
1.IVC filters: Do current data support the indications, John Kaufman, MD. Endovascular Today. February 20102 2.Guidelines for the use of retrievable and convertible vena cava filters: Report from the Society of Interventional Radiology Multidisciplinary Consensus Conference. Journal of Vascular and Interventional Radiology 2006 3.Update on Inferior vena cava filters. Thomas B. Kinney. Journal of Vascular and Interventional Radiology, 2003.



